Introduction
As far as the health of human bone is concerned, Ca is the most important mineral at least in quantity because the vast majority (>99%) of total body Ca is present in the bone [1]. It was reported that 67.4% of the Koreans could not meet a minimum daily Ca requirement, 700–1,000 mg [2]. The large amounts of Ca requirement are probably due to very low rate of Ca absorption in the small intestine [3]. Several materials that promote Ca availability in the intestine have been identified and been commercially available. One of them is casein phosphopeptides (CPP), which readily makes Ca soluble in the small intestine when free Ca is bound to CPP [4]. A CPP could, therefore, help a Ca absorption in the small intestine. CPP is a collection of trypsin-digested sodium caseinates, and active components are present in casein-hydrolyzed peptides by trypsin. These active components are one or more phosphorus linked to serine. Typically, active components make up 16%–25% of CPP, depending on the preparation methods, and the rest is phosphorus-lacking peptides.
For the design of healthy foods, food scientists select and add health-promoting materials to desirable processed foods. Several of health promoting materials range from nutrients as basic as vitamins, and minerals to functional ingredients such as docosahexaenoic acid, ω-3 fatty acid, CPP, etc. A good many of process foods with the specific bioactive ingredients added can be widely found in markets. One example is a frozen pizza produced in domestic food manufacturer, Ottogi, with CPP and calcium fortified. One of the issues that concerns us is to evaluate and determine actual amounts of functional ingredients remained in the final products. Act on labelling and advertising of foods in Korea [5] stipulates that instead of a product name, a component name of ingredients added to foods can be written on food package only if it can be quantified. Although the accurate amounts of every ingredients are carefully weighted and poured into one batch and products are manufactured, target values may not be met. Gaps between target and actual values can be ascribed to uneven distribution of functional ingredients due to the very small amounts added, and some loss on measuring cups and manufacturing utensil. Another challenge that complicates the measurement of target materials would be the lack of analytical method. When a target material is present in a relatively very simple system, well-defined method is available. However, if the same material is present in a very complex food system, the same method would not work because the material to be determined and other ingredients of no interest share similar properties that are critical to measuring the content of the target of concern.
In our study, Dangmyon (starch vermicelli) is chosen to which Ca and CPP were added. Dangmyon, also known as a glass noodle, is a traditional starch noodle, made of starch from sweet potato, H2O, and thickening agents [6]. The major dishes whose ingredient is Dangmyon include Japchae, dumpling, and fried seaweed. These dishes are very popular with most people, and Dangmyon could be a better Ca carrier for those who may be Ca-deficient. In addition, its composition is so simple that its analysis of CPP would be feasible. Industrial-scale of Dangmyon was manufactured aiming for the indirect quantification of CPP. The active components of CPP are phosphopeptides [4]. From compositional view point, a single ideal CCP molecule possesses 3 phosphorus (P), and P content of 100 g CPP powder is estimated to ~800 mg [7]. We hypothesize that CCP content in the Dangmyon would be indirectly quantified on the basis of phosphorus contents in the CPP powder. Method for measurement of CPP in a CPP powder or dairy foods such as cheese is well established, but when it is present in a complex food system, no direct method is available probably due to inability of CCP to be separated from the rest of components. In this paper, attempt made was reported to indirectly measure CPP contents in a Dangmyon on the basis of P content in a CPP.
Materials and Methods
Two kinds of Ca sources used were egg shell powder (NewCal, Poonglim Foods, Korea) and milk Ca (Trucal milk calcium complex, Granbia, USA). Casein phosphopeptides (CH-CPP20, Choheung, Korea) were prepared according to methods by Choi et al. [4], and available in market under the brand name of CH-CPP20. The detailed information, i.e., brand name and manufacturer, on ingredients for the preparation of Dangmyon, which Woojung Foods possesses, cannot be disclosed. All chemical reagents were AOAC grade.
It was a challenging task to determine an optimum dose of Ca added to Dangmyons. However, the safe maximum levels of minerals fortified to foods have been set up per 100 kcal portion based on tolerable upper intake level, the proportion of fortified foods in diets of individuals, etc. [8]. According to this model, the upper limit of Ca level was estimated to be 191 mg per 25 g (one serving size) of Dangnyon. The target level of Ca was determined subjectively considering several factors such as cost, sensory on the finished Dangmyon, and the physical properties of Dangmyon and Ca sources. The target levels are shown in Table 1.
Industrial-scale facilities in Woojung Foods were used to manufacture all Dangmyons used in this study. The detailed manufacturing process and the formulation are the proprietary information that belongs to Woojung Foods, and cannot be disclosed. The very limited formulation, therefore, was shown in Table 1, and the brief outlines of manufacturing process were presented as follows. A 10 kg of sweet potato and a 200 kg of water were weighted into a mixing tank that was cooked at 60°C–70°C with a mild agitation before gelatinization. The rest of starch (340 kg) was put into the tank, well mixed for 20 min, and vacuumed at 70–75 mm Hg for a removal of air. The cooked starch was pushed to a cylindrical extruder with a 12 mm diameter opening to make Dangmyon. Dangmyons were then immersed in boiling water of ~95°C for 30 s, cooled in cold water (13°C), aged for a few hr at room temperature, frozen at –5°C to –15°C for 24 hr, thawed with cool water, and finally dried. Final moisture content was assumed to be ~13%. The Dangmyons were cut into 30 cm long and packed.
Moisture, fat, and total protein were determined as described by Marshall [9]. For Ca and P analyses, samples ashed at 600°C were solubilized with 2% HNO3, which was then fed into inductively coupled argon plasma emission spectroscopy. Wavelengths used for Ca and P were 317.9 and 214.9 nm, respectively. Amylose content of samples was measured, as described by Zhu et al. [10]. 40 mg of amylose (CAS # 9005-82-7, Sigma-Aldrich, USA) was weighted into a 100-mL volumetric flask to which 1 mL of 95% ethyl alcohol and 9 mL of 1 M NaOH were added. The flask was heated in water bath to completely dissolve amylose. The clear amylose solution was made to 100 mL with distilled water. This was serially diluted to construct standard curve with distilled water from 8% to 40% of amylose solution. The amounts of acetic acid (1 N) added to each standard solution as well as sample solution were in proportion to the amounts of 1 M NaOH. 100 mg of Dangmyon was taken and treated similarly to the preparation of standard solution. 2 mL of iodine solution (2.0 g of KI and 0.2 g of I2 diluted to 100 mL with distilled water) was pipetted to each standard and samples, which was then made to 100 mL with distilled water. The final solution was allowed to fully develop the color for 30 min. Absorbance was measured at 620 nm.
A 200 mL of boiling water was transferred to 500 mL media bottle placed on hot plate, to which 20 g of 5-cm Dangmyon was put. It was boiled for 8 min, immediately removed to a cool media bottle with 200 mL of cool water in it. After 1 min, cool Dangmyon was removed onto funnel lined with Whatman filter paper #4. Water was allowed to be drained, which was used for determination of chemical composition.
Two sources of Ca, egg shell (egg) and milk Ca, were used for the fortification of Ca to Dangmyon with one and two levels, respectively, which formed three replicates. Their effects on three different sampling stages were F-tested. Cooking effect on Ca, P, ash, and protein contents were similarly tested after all values were converted on dry matter basis. Statistical significance was declared at p<0.05 (MS Excel, 2013).
Results and Discussion
Chemical composition of major ingredients necessary to manufacture Dangmyon was summarized in Table 2. Ca and P contents of sweet potato were 74.86 and 14.49 mg/100 g, respectively. The contents of Ca in egg and milk Ca, which were used for fortification of Ca to Dangmyon, were 39 and 25 g/100 g, respectively, which are well above or meet their minimum limits. Phosphorus contents of two sources were 0.129 and 14 g/100 g, respectively. CPP was found to have 105 mg of Ca and 754 mg of P per 100 g. The P in CPP is relatively constant, and an active component that plays a pivotal role in solubilization of Ca in the intestine. Our hypothesis was that P content in CPP is 6 times more than in egg Ca, and could be used as an indirect measurement of CPP content present in Dangmyon. It has been reported that the whole casein micelles possess ~800 mg of P per 100 g protein on dry matter basis [7], which our value for P content in CPP was in agreement with. Milk Ca showed the highest P content among principal ingredients.
Our target Ca content was set to be between 60 and 125 mg per 25 g of Dangmyon. The amount of CPP fortified to Dangmyon was determined as follows: we assumed that a half of total Ca was adsorbed naturally and CPP can then help adsorb the remaining Ca. 30% of CPP is required for the amounts of Ca, and purity of CPP (Ch-CPP20) is ~22% [4], on the basis of which the amount of CPP was calculated. Based on Ca content of each ingredient, the amounts of egg and milk Ca to be added were obtained, and the final recipe appears in Table 1. Since sweet potato does have endogenous Ca, this was taken into account when formulation was made. Sweet potato constitutes more than 98% of Dangmyon, and its composition determines that of Dangmyon. This was confirmed in Tables 1 and 3. Dangmyons manufactured in Woojung Foods were purchased at local stores, which served as control (CON). These could have theoretical value of 18.7 mg Ca and 3.6 mg P.
Dangmyon was manufactured by batch type of extrusion method. Three samples were taken during production of Dangmyon, the beginning, middle, and near end. Since Ca and CPP-added Dangmyon had higher protein content than one not fortified (CON), protein and moisture contents were measured, by which the uniform distribution of Ca and CPP during production of Dangmyon was indirectly evaluated. F-test was made for the effect of sampling stage on moisture and protein, and it was found that there were no statistical differences (p<0.05) among sampling stages (Table 4). Moisture content was ~10%, which was a little lower than that of sweet potato. According to Woojung Foods, 100 kg of sweet potato usually gives ~100 kg of Dangmyon since moisture content of Dangmyon and sweet potato is similar. From now on, samples taken in the middle of production were used for further experiment. It should be kept in mind that CON was purchased at local stores instead of manufacturing and thus no samples were taken at different stages.
Dangmyon is widely used for making several dishes such as Japchae, and a boiling process is an essential step before any dishes are made. Ability of Dangmyon to retain Ca and CPP after boiling was therefore investigated. Fig. 1 shows changes in moisture content that increased by 7 times as much as one not cooked. Similar results were reported by Ko & Kim [11]. An increase in moisture content at cooking was accompanied by a drastic decrease in Ca and P contents in Dangmyon. For fair comparison before and after cook, values for Ca, P, ash, and protein were converted on the dry matter basis (Table 5). F-test was made to see cooking effect on loss of components and it was found that no loss occurred at p<0.05. In Table 5, individual data as well as means of three were reported. Ca and P contents in cooked Dangmyon were a little higher than in no cooked one, but ash and protein contents showed reverse trend, although these differences were not statistically different at p<0.05.
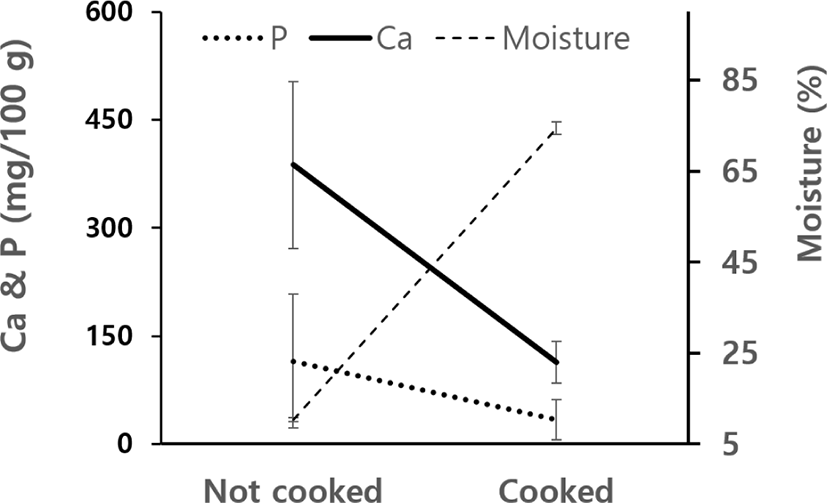
Table 3 presented chemical composition of Dangmyon fortified with two Ca sources and CPP according to the recipe appeared in Table 1. Target and experimental vales for Ca in CON were 18.7 and 24 mg per 25 g of Dangmyon, respectively. This difference could be due to the fact that moisture content of Dangmyon was assumed to be ~13% at the time of writing the recipe in Table 1. In fact, moisture content turned out to be a slightly lower value of ~10%. Careful comparison of Tables 3 with 1 indicates that the target values for Ca were just satisfied. The set values for egg Ca, milk Ca-high, and milk Ca-Low were 126.0, 98.6, and 60.6 mg per 25 g of Dangmyon, respectively. The corresponding actual values were 125, 98, and 67 mg of Ca, which are close to set values. Values for moisture, ash, and protein agree well with those reported by Jeon et al. [6] except amylose content. Our amylose content was higher than most reported values by Han et al. [12]. The reason was not clear.
Walk-through exercises for the estimation of CPP in three trials were shown in Table 6. Example for estimation of CPP present in Dangmyon with Egg Ca is given here. Ingredients that can contribute only to P content were listed in leftmost column. CON represents Dangmyon bought at local stores and served as control. Its P content was an analyzed value that is same as bottom line of Table 3. This value (16.39 mg) was used as P content in sweet potato when partition of total P was made into three ingredients. The P (1.43 mg) in each Ca source was calculated by multiplying P values (129.7 mg/100 g) taken from Table 1 by its amounts (1.097 g) added to 100 g Dangmyon. The P content in CPP was the difference between total P (19.83 mg) and sum (16.39+1.43) of those two P values above. Then, P value (2.02 mg) assigned to CPP was converted into amounts of CPP (0.266 g) by multiplying 2.02 mg by CPP amounts (0.132 g) per mg of P. The amount of CPP (0.132 g) corresponding to 1 mg P was obtained by dividing 100 g of CPP by its P content (754.86 mg). It was found that this method underestimated its CPP content to 0.266 g per 100 g Dangmyon although 0.301 g per 100 g Dangmyon was added. Similar calculation was made to Dangmyon with high and low milk Ca. It was found that this method extremely underestimated the amount of CPP in high milk Ca to be 36.4%. In low milk Ca, this highly overestimated its CPP content three times more. This is probably ascribed to the fact that too much content of P in milk Ca hindered the precise partition of P into CPP. Therefore, when Ca source is considered, Ca source with very low concentration of P content must be selected.
Conclusion
Phosphorus content in CPP is relatively constant, and higher compared to that in egg Ca. This could be exploited to indirectly measure CPP content in Dangmyon fortified with Ca and CPP. Partition of total P into sweet potato, egg Ca, and CPP was made, and P value assigned to CPP was changed to CPP content. When P content is very low such as egg Ca, this method worked well, although a little underestimated. However, when Ca source had too much P content, this method did not work out. Therefore, if CPP measurement is essential, then Ca source should be chosen to be very low in P content. Dangmyon is used as a material for popular dishes, Japchae. One of the essential processes is a boiling step for 8 min. It was found that there was no loss of Ca and CPP after boiling. Therefore, Dangmyon could be an excellent carrier of Ca for humans.






