Introduction
Modern agribusiness and food processing industry have dramatically altered the pattern of nutrients and lifestyles in the human diet (Uauy et al., 2001; Sim et al., 2010). Public awareness of the crucial link between nutrition and health is growing rapidly, especially in the role that diets play in combating age-associated chronic diseases, such as cardiovascular disease, malignant disease, diabetes, osteoporosis, arthritis, and Alzheimer's disease (Heird et al., 1997; Tapiero et al., 2002). Research conducted over the past fifteen years has shown that dietary fat is more than just fuel, but that it is an essential nutrient that influences every aspect of physiological and metabolic processes of human body (Liang et al., 2008). Eating a healthy balanced food will reduce risk of a host of diseases. It might even save one’s life. Essential fatty acids must be supplied from foods we eat to sustain human-gene expression and overall metabolic homeostasis (Koo, 2003; Gleissman et al., 2010). Today’s industry and consumers alike are searching new food fat sources supplying both long chain omega-3 and omega-6 polyunsaturated fatty acids in an adequately balanced ratio, ideally 1:1 (Takeuchi et al., 2008; Sim et al., 2010). Chinese diets have been rapidly changing with remarkably increased consumption of plant oils, from 14 kg per capita in 2004 to 17 kg per capita in 2007 (Sim et al., 2010). Chinese diets are deficient in omega-3 fatty acids, and have excessive amounts of omega-6 fatty acids compared with the diet on which human beings evolved and their genetic patterns were established (Liang et al., 2008; Sim et al., 2010). Excessive amounts of omega-6 polyunsaturated fatty acids (PUFA) and a very high omega-6/omega-3 ratio, as is found in today's Chinese diets, promote the pathogenesis of many diseases, including cardiovascular disease, cancer, and inflammatory and autoimmune diseases, whereas increased levels of omega-3 PUFA (a low omega-6/ omega-3 ratio) exert suppressive effects (SanGiovanni and Chew, 2005; Mita et al., 2010; Cockbain et al., 2012). In the secondary prevention of cardiovascular disease, a ratio of 4/1 was associated with a 70% decrease in total mortality (Kris-Etherton et al., 2002). A ratio of 2.5/1 reduced rectal cell proliferation in patients with colorectal cancer, whereas a ratio of 4/1 with the same amount of omega-3 PUFA had no effect (Sim et al., 2010). The lower omega-6/omega-3 ratio in women with breast cancer was associated with decreased risk (Simonsen et al., 1998). A ratio of 2-3/1 suppressed inflammation in patients with rheumatoid arthritis, and a ratio of 5/1 had a beneficial effect on patients with asthma, whereas a ratio of 10/1 had adverse consequences (Sim et al., 2010).
Hence, the objective of this research project is to collect local oil grain samples, to determine their physicochemical properties and lipid content, and finally to subject them to GC analysis for their fatty acid compositions. The presence and absence of omega-3 fatty acids and the degree of omega-3 fatty acid are to be suggested as new oil sources for the supply of dietary omega-3 fatty acid in China including finding out the usability for functional diary foods.
Materials and Methods
Situated in the east of Jilin Province, Yanbian Korean Autonomous Prefecture is bordered by Russia in the east, and is separated by the Tumen River from North Korea. Purchased oil grain seeds (S, F, P, SB, and C) from West Market that is the most famous market in Yanji City (Fig. 1). And then, materials were dried at YUST which is short for Yanbian University of Science and Technology.
Weighed 10 g of five oil grain seeds respectively and counted the quantity. Afterwards, weighed one spoon of each samples by three times and counted them.
Before lipid extraction, five samples (S, F, P, SB, and C) with equal dried condition should be put into drying oven, and then were ground using an electronic grinder. Total lipids were extracted from S, F, P, SB, and C with chloroform: ethanol (2:1, vol/vol) by the Folch method.
One gram of sample were weighed into a test tube with 20 mL of chloroform: ethanol (2:1, vol/vol), and made sure it was homogenized well and well sealed. It was left overnight extraction to separate into phases. The homogenate was filtered through a Whatman filter paper into a 100-mL graduated cylinder, and filtered it with another 10 mL Folch’s solution plus 4 mL DDW twice, and shook it well. It was left overnight to separate into obvious phases. After phase separation, the volume of the lipid layer was recorded, and the top layer was completely siphoned off, which contains water soluble and alcohol soluble substances. The volume of chloroform soluble lipids (bottom phase) was recorded as V mL. Lipid contents were determined gravimetrically for S, F, P, SB, and C. Two mL of bottom layer was taken on a pre-weighted Aluminum foil (A0 g). Then it was dried under room temperature. Weighed the dried aluminum foil and recorded it as A g.
Calculation method: (A—A0) g/2 mL × V mL = Total extract yield = E mgE g / 1 g sample = % of fatty acid yield from sample
The analysis of fatty acid composition and omega-3 fatty acid content of oil grain seeds was performed on an Agilent 6890 gas chromatograph (Agilent Technologies, Santa Clara, CA, USA) equipped with a flame ionization detector (FID) and fitted with a fused silica capillary column (DB-23, 60 m × 0.25 mm; film thickness 0.25 μm; J&W Scientific Co, Folsom, CA, USA). Helium gas was used as a carrier with a flow rate of 2.0 mL/min, and a split ratio of 20:1. The temperature of the inlet and the FID were 260℃ and 270℃, respectively. The sample size was 1 μL.
Results and Discussion
Weigh 10 g of oil grain seeds respectively and count the number, then come out to the quantity of samples per 10 grams, namely average (Table 1). Because of different sizes of oil grain seeds, of course, equal weight of samples have different quantities. Peanut is bigger than others so 13 pieces of peanut is about 10 g. Similarly, 10 g of sesame seed with more quantity is 349. Quantity from largest to smallest order is sesame (S), flax (F), soybean (SB), corn (C), and peanut (P).
| Oil grain seeds | Tsp | 10 g | |
|---|---|---|---|
| NO./Tsp | g/Tsp | No./10 g | |
| Sesame | 1,145 | 3.28 | 3,491 |
| Flax | 416 | 2.28 | 1,823 |
| Peanut | 7 | 5.63 | 13 |
| Soybean | 22 | 4.46 | 50 |
| Corn | 8 | 2.55 | 31 |
Even though table spoon method is much more available naturally, it is not easy to count. Therefore, consumers need a simpler and more convenient method.
According to report of adequate intake levels for linolenic acid by the institute of medicine at the national academy of sciences in 2002 (Pan et al., 2012), 1.6 grams per day were recommended for male teenagers and adult men, the recommended amount were 1.1 grams per day for female teenagers and adult women.
It could be figured out that 2.99 grams flax contains 1.6 grams omega-3 fatty acids. Also it can be calculated that 2.99 grams flax are equal to 544 pieces flax and 1.3 table spoon (more than 1 spoon) flax. And the female daily intake 2.06 grams (375) flax or 0.9 table spoon (about 1 spoon) flax is equal to 1.1 grams omega-3 fatty acids.
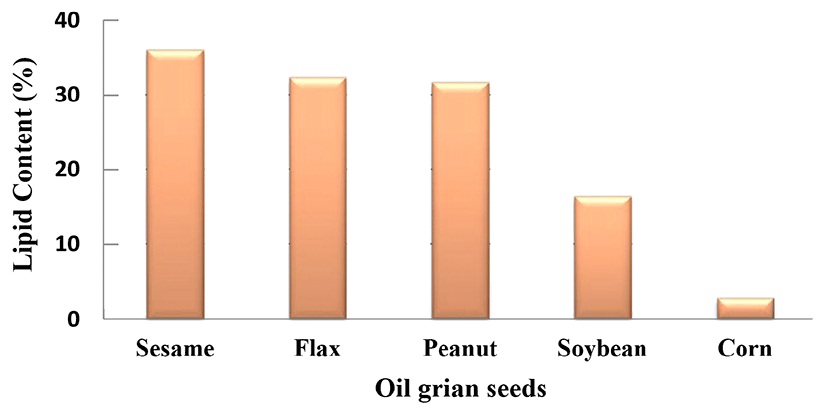
According to Fig. 2, the average of lipid component of sample S as 36.13%, F as 32.43%, and P as 31.75% are more than 30%, but SB as 16.47% and C as 2.93% are less than 30%.
In general, the ω-6 and ω-3 PUFAs when consumed in the form of dietary triglyceride from various food sources undergoes digestion in the small intestine which allows for absorption, transport in the blood, and subsequent assimilation within tissues themselves through the body (Tekeuchi et al., 2008; Mita et al., 2010).
The ratio between omega-6 and omega-3 fatty acids was very important for human’s health. According to Table 2, flax contains the highest omega-3 FA and others are very few, including corn least. However, omega-6 FA content is much higher than omega-3 FA, including S, P, SB exceeded 40%, and even SB is nearly to 50%. By contrast, flax contains the least omega-6 FA, which is only 16.86%. Also among the content of SAFA, MUFA and PUFA, corn contains highest content of SAFA, which is more than four times of flax. S and P contain more than 40% of the MUFA (Table 2). Hence, it could be explained that omega-3 FA influence the content of PUFA. In general, PUFA is proportional to omega-3 FA.
Recently, with the development of upgrading life style, the previous balanced diet could be broken. It could result many modern diseases such as cardiovascular disease, high blood pressure, high blood cholesterol, and so on. Namely, owing to the imbalanced ratio between omega-6 and omega-3 fatty acids from the eating habits of life, the number of patients would be dramatically increasing.
Also omega-6 and omega-3 ratio are presented. F is the maximum, and then is SB, S, and P. Corn has the smallest proportion (Table 2).
Therefore, eating more flax can help people balance the body of omega-6 and omega-3 ratio and make people healthier. People know that the optimum ratio is 1:1, while the omega-6 and omega-3 ratio of the modern body even up to 20:1. Hence, people try to reduce the proportion of 1:1. According to result of this study, when people intake the same amount of flax and corn, it could be readjusted the ration of the omega-6 and omega-3 is to 1:1. Flax contains 16.86% omega-6 fatty acid and 53.43% omega-3 fatty acid. There is 37.57% omega-6 fatty acid in corn, Whereas omega-3 fatty acid does not exist in corn. Omega-3 FA adds up to 54.43% between flax and corn. And the sum of omega-6 FA is 53.43%.
Conclusions
The health benefits of the omega-3 fatty acids continue to grow, but there is a troubling trend worldwide. Most individuals are not consuming enough of these fatty acids for optimal health. Yet omega-3 fatty acids are essential for all stages of life from birth through adulthood. Moreover, today’s consumers are far more conscious and informed of the link between health and foods than previous generations, and this change in attitude has an increasingly significant impact on their food choices. Following the trend in the West, the Chinese consumers also began to search for foods rich in omega-3 fatty acids and antioxidants. Not too many omega-3 fatty acids including oil grains seeds are qualified. Therefore, combination of Chinese grown flaxseed and corn may be the best option to secure good dietary supply of omega-3 fatty acids in China. Furthermore, this study could be suggested that omega-3 fatty acids extracted from various oil grains could be used for improving the quality of various dairy foods.